  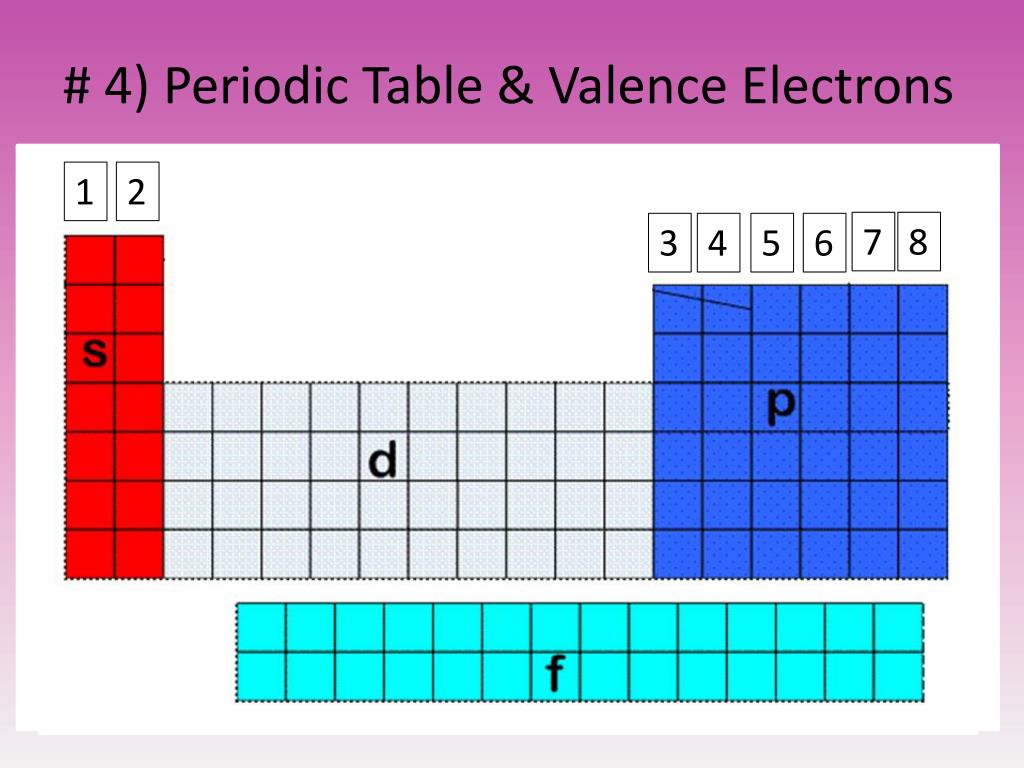
While arranging the elements according to their atomic weight, if he found that they did not fit into the group he would rearrange them. In 1869, Russian chemist Dmitri Mendeleev created the framework that became the modern periodic table, leaving gaps for elements that were yet to be discovered. He arranged the elements in eight groups but left no gaps for undiscovered elements. He found that every eight elements had similar properties and called this the law of octaves. For example, carbon, being12 times heavier than hydrogen, would have an atomic weight of 12.īritish chemist John Newlands was the first to arrange the elements into a periodic table with increasing order of atomic masses. 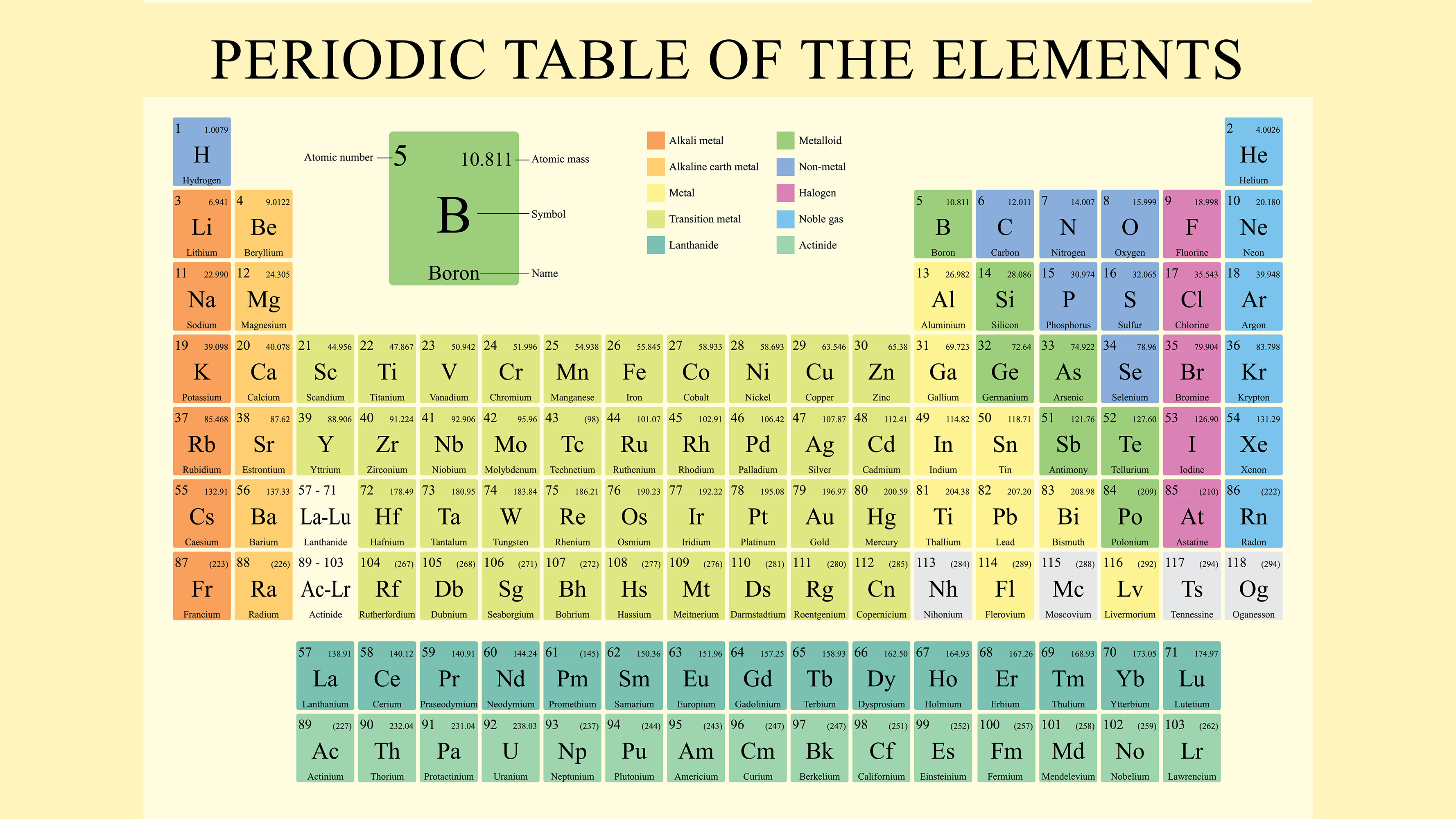
They concluded that hydrogen would be assigned the atomic weight of 1 and the atomic weight of other elements would be decided by comparison with hydrogen. He arranged them in groups of three in increasing order of atomic weight and called them triads, observing that some properties of the middle element, such as atomic weight and density, approximated the average value of these properties in the other two in each triad.Ī breakthrough came with the publication of a revised list of elements and their atomic masses at the first international conference of chemistry in Karlsruhe, Germany, in 1860. Forty years later, German physicist Johann Wolfang Döbereiner observed similarities in physical and chemical properties of certain elements. In 1789, French chemist Antoine Lavoisier tried grouping the elements as metals and nonmetals. Elements in Group 2 have similar chemical properties, they are called the alkaline earth metals.Among the scientists who worked to created a table of the elements were, from left, Antoine Lavoisier, Johann Wolfang Döbereiner, John Newlands and Henry Moseley. GroupĮlements in Group 1 (periodic table) have similar chemical properties and are called alkali metals. The vertical columns have similar properties within their group for example Lithium is similar to sodium, beryllium is similar to magnesium, and so on. The table below shows that elements increase from left to right accordingly to their atomic number. The periodic law is found to help determine many patterns of many different properties of elements melting and boiling points, densities, electrical conductivity, reactivity, acidic, basic, valance, polarity, and solubility. Thus, we can see that elements are arranged based on their atomic number. 39.1, respectively), yet argon is in front of potassium. As mentioned before, argon weights more than potassium (39.9 vs. Similar properties recur periodically when elements are arranged according to increasing atomic number."Ītomic numbers, not weights, determine the factor of chemical properties. With Moseley's contribution the Periodic Law can be restated: Moseley was able to tie the X-Ray frequencies to numbers equal to the nuclear charges, therefore showing the placement of the elements in Mendeleev's periodic table. Moseley did researched the X-Ray spectra of the elements and suggested that the energies of electron orbitals depend on the nuclear charge and the nuclear charges of atoms in the target, which is also known as anode, dictate the frequencies of emitted X-Rays. Elements were placed into groups that expressed similar chemical behavior. An example of this was with argon (atomic mass 39.9), which was put in front of potassium (atomic mass 39.1). Explaining that a given set of properties reoccurs every eight place, he named it the law of Octaves.Ītomic Number as the Basis for the Periodic LawĪssuming there were errors in atomic masses, Mendeleev placed certain elements not in order of increasing atomic mass so that they could fit into the proper groups (similar elements have similar properties) of his periodic table. R Newland arranged the elements in increasing of atomic weights. This gave scientists the tools to reveal the relationships between elements. In 1859 two physicists Robert Willhem Bunsen and Gustav Robert Kirchoff discovered spectroscopy which allowed for discovery of many new elements. However, at the time of this discovery too few elements had been discovered and there was confusion between molecular weight and atomic weights therefore, chemists never really understood the significance of Doberiner's triad. By 1829, chemist Johann Wolfgang Doberiner observed that certain elements with similar properties occur in group of three such as chlorine, bromine, iodine calcium, strontium, and barium sulfur, selenium, tellurium iron, cobalt, manganese. Around the same time, two chemists Sir Humphry Davy and Michael Faraday developed electrochemistry which aided in the discovery of new elements. In 1804 physicist John Dalton advanced the atomic theory of matter, helping scientists determine the mass of the known elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
